Do horses in training need regular deworming?
Words - Professor Jacqui Matthews BVMS PhD FRSE FRCVS - RCVS Recognised Specialist in Veterinary Parasitology / Director of Veterinary Science, Austin Davis Biologics
With wormer resistance increasing and no new products expected soon, targeted parasite control is now essential for all horses. This approach combines risk assessment, diagnostic testing, and paddock management to reduce infection levels and ensure only those horses that need treatment are wormed. This article outlines practical strategies for sustainable worm control in horses in training.
Introduction
Almost every horse will encounter parasitic worms in its life. While it is unnecessary to completely eradicate worms from groups of horses, larger burdens can lead to disease, especially in young or older individuals. Traditionally, control involved giving wormers to all horses in a group without considering individual requirements or risks. This one-size-fits-all approach has contributed to the development of resistance to wormers (anthelmintics) over the past forty years. Today, these medications are less effective against all major types of worms and cases of multi-drug resistance are on the rise, posing serious challenges for the future control of these important pathogens. With no new wormers expected in the foreseeable future, maintaining the effectiveness of the current anthelmintics is essential.
Parasites of concern
Small redworms (cyathostomins) and tapeworms (Anoplocephala perfoliata) are the most important parasites affecting horses. Even horses in training, with limited grazing, can be exposed to these infections. Young horses (≤2 years), particularly those recently arrived from breeding farms, often carry higher parasite burdens. These animals are of particular concern, as breeding farms are commonly where wormer resistance first emerges, meaning new arrivals may introduce parasites that are more difficult to treat.
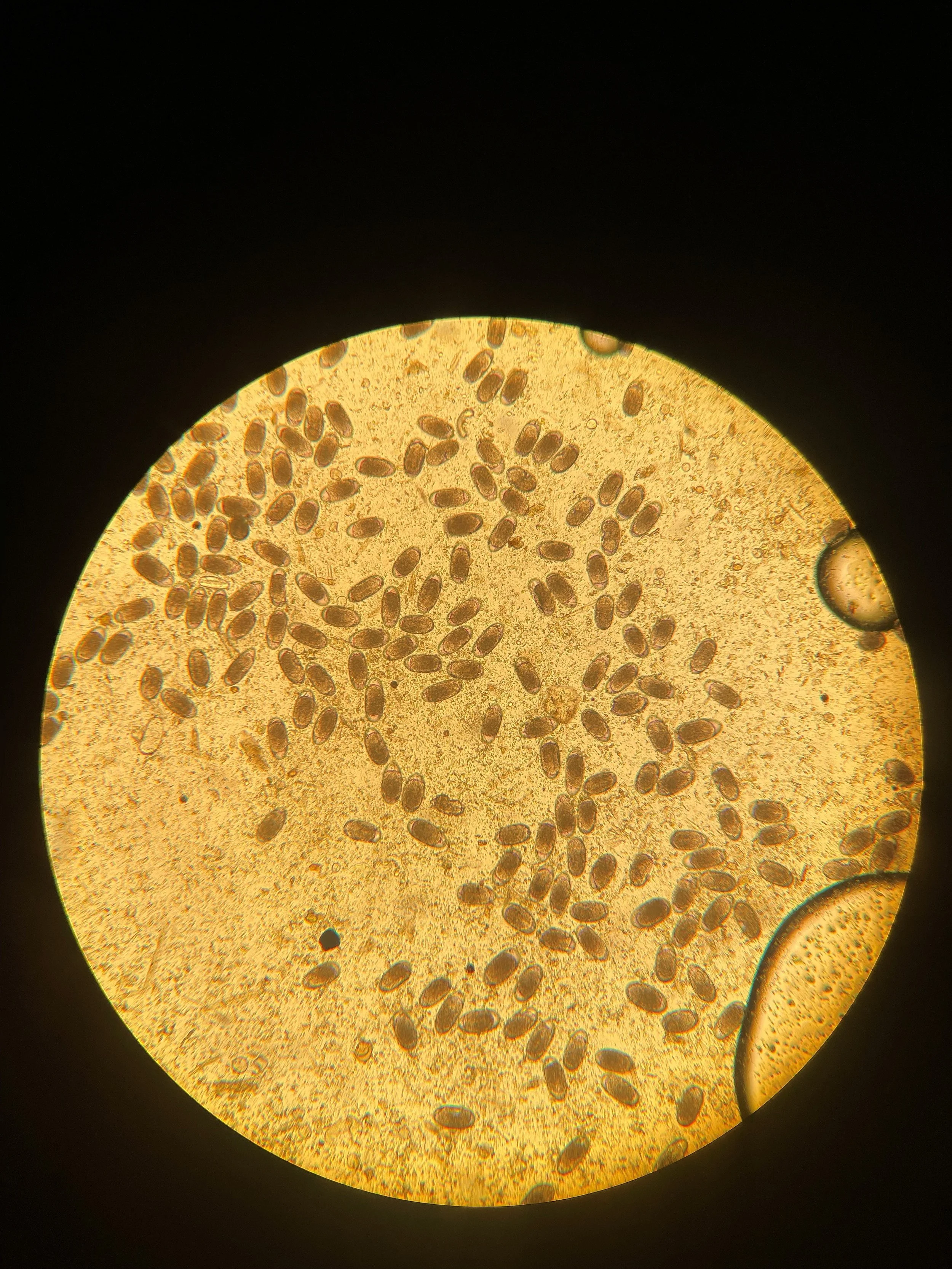
Fig. 1
Nearly all horses are infected with small redworms at some point. Most infections are sub-clinical, but heavy burdens of encysted larvae in the intestinal wall (Fig. 1) can cause weight loss, diarrhoea, colic and, in severe cases, life-threatening larval cyathostominosis. Outbreaks of this disease have been reported on farms where Thoroughbred yearlings were grazed on poorly-managed paddocks (Byrne et al. 2025).
Encysted larvae are less susceptible to most wormers, with only moxidectin and a five-day course of fenbendazole licensed in the UK/EU for activity against all stages. However, fenbendazole resistance is now widespread, and reduced effectiveness of moxidectin is increasingly reported (Nielsen 2022), making it essential to preserve these treatments for horses with significant burdens.
In most healthy adult horses, particularly those in training with no/limited grazing or turned out to well-managed paddocks, small redworm burdens are generally low, with only a small proportion of individuals carrying higher infection levels. While the impact of subclinical infections on performance remains unclear, it is likely that higher parasite burdens are more likely to have negative effects.
The common equine tapeworm, Anoplocephala perfoliata, can affect horses of all ages and may cause colic even at relatively low burdens; as few as 20 worms can damage the intestine, particularly at the small–large intestinal junction (Matthews et al. 2024). Infection levels vary between groups, but tapeworms are especially common on breeding farms. Their life cycle involves soil-dwelling mites as intermediate hosts, making control challenging as these mites can spread infection around paddocks and harbour infective stages for prolonged periods. There is also growing concern about reduced effectiveness of the two wormers licensed for tapeworm treatment.
Other worms infect horses but are generally less relevant on training yards. Ascarids mainly affect foals (4–8 months), though older or immunocompromised horses can be infected. Pinworms can cause tail itching, signalling hygiene issues. The large strongyle, Strongylus vulgaris, is generally rare but can cause severe colic or peritonitis and should be monitored in high-risk groups on poorly managed paddocks.
Resistance to wormers
A range of wormers is available for horses, including benzimidazoles (fenbendazole), tetrahydropyrimidines (pyrantel) and macrocyclic lactones (ivermectin, moxidectin) for small redworms, ascarids, and other roundworms, and praziquantel, which specifically targets tapeworms.
Resistance to these drugs is increasingly reported. Small redworms are of particular concern: benzimidazole resistance is widespread, pyrantel resistance is likely and reports of reduced effectiveness of macrocyclic lactones are growing (Nielsen 2022). In ascarids, resistance to macrocyclic lactones is common, with reduced sensitivity to benzimidazoles and pyrantel occasionally reported.
More recently, reduced effectiveness of double-dose pyrantel and praziquantel against A. perfoliata was reported in the US (Nielsen 2023), with anecdotal cases emerging in Europe. For horses in training, these trends are important. Even with limited grazing, they can pick up worms at turnout, on training tracks or while grazing paddocks during rest periods. Furthermore, new infections, including resistant parasites, may be introduced by horses coming from breeding premises.
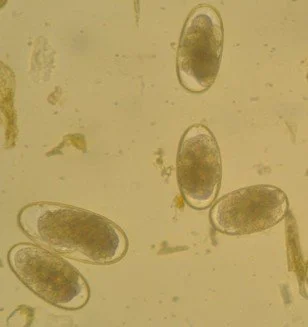
Multiple strongyle eggs in FEC
If resistance continues to rise, heavy parasite infections will become increasingly difficult to treat. This makes it essential to avoid unnecessary worming and to base treatments on risk assessment and testing.
Risk assessment
Effective parasite control begins with a thorough assessment of infection risk at both the individual and group level. This allows targeted interventions to be implemented and their effectiveness monitored over time. Online tools such as WhatsYourWormRisk.com provide a structured way to assess risk profiles and support evidence-based decision-making. Key factors influencing risk include age, grazing exposure, pasture management and yard biosecurity. Most adult horses (>4 years) develop some immunity, reducing their susceptibility to high worm burdens, while younger horses are more likely to carry heavier infections and are therefore at higher risk.
Older horses (>20 years), particularly those with PPID, may also be more vulnerable. Regular risk assessment with a veterinary surgeon helps trainers identify management practices that could inadvertently increase infection risk. Even short daily turnout on paddocks without dung removal, or shared grazing during rest periods, can facilitate parasite transmission. Horses kept as companions under less intensive management can also act as sources of infection. A critical part of any assessment is reviewing recent test results, which can reveal whether the estimated risk matches reality; for example, multiple horses present with high faecal egg counts (FECs) despite a presumed low-risk status.
Parasite testing
Diagnostic testing is central to parasite control, providing objective data to guide treatment decisions and reduce unnecessary wormer use. By identifying which horses excrete worm eggs or carry parasite burdens, trainers can target treatments more effectively while reducing selection pressure for anthelmintic resistance. The tests available include FECs and antibody-based assays for tapeworms and small redworms.
Fig. 2
FECs estimate the number of parasite strongyle (Fig. 2) or ascarid eggs a horse is shedding in dung, providing insight into its potential to contaminate the environment. In horses in training, FECs should be performed every 12–24 weeks as determined by veterinary risk assessment. Egg shedding is uneven between horses: in low-risk groups, fewer than 20% of horses account for ~80% of eggs excreted, while many shed a few/no eggs and do not require treatment. Treatment thresholds usually fall between 200-500 eggs per gram. FECs only detect eggs from adult females and provide no information on male or immature/encysted stages, so they are not a complete measure of parasite burden. If several horses show unexpectedly high FECs, this may indicate a management issue (access to contaminated paddocks, emergence of resistance). FEC reduction tests can help assess whether wormers are effective by comparing egg counts before and 2 weeks after treatment.
FECs have low sensitivity for detecting tapeworms. Infections are better assessed using saliva or blood tests which measure A. perfoliata-specific antibodies that correlate with tapeworm burden level (Lightbody et al. 2016).
Testing can be carried out at any time of year. In horses with access to paddocks, spring testing is useful to identify infected horses that will shed eggs in spring/summer when intermediate host mites are most active (Wickenden et al. 2025). End-of-summer testing is important for horses that have grazed during a rest period, as it can identify those that have acquired tapeworm burdens that put them at risk of colic. Testing can be done three months after the last treatment with saliva tests or four months with blood tests.
FECs do not reliably reflect small redworm burdens, particularly encysted larvae. Instead, the small redworm blood test is available that measures antibodies to all parasite stages, including encysted larvae (Lightbody et al. 2024). In horses in training, this test can be performed at any time of year, but is especially useful when considering encysted larvae treatments in autumn/winter.
This test can help avoid unnecessary blanket treatments. For example, in a cohort of over 900 sport horses, more than 80% had small redworm blood test results below the 10,000-worm threshold, and over 60% were below the 1,000 worm-threshold, indicating very low parasite levels (Matthews et al. 2024). Using the test in such situations can therefore significantly reduce treatment frequency compared with worming the entire group.
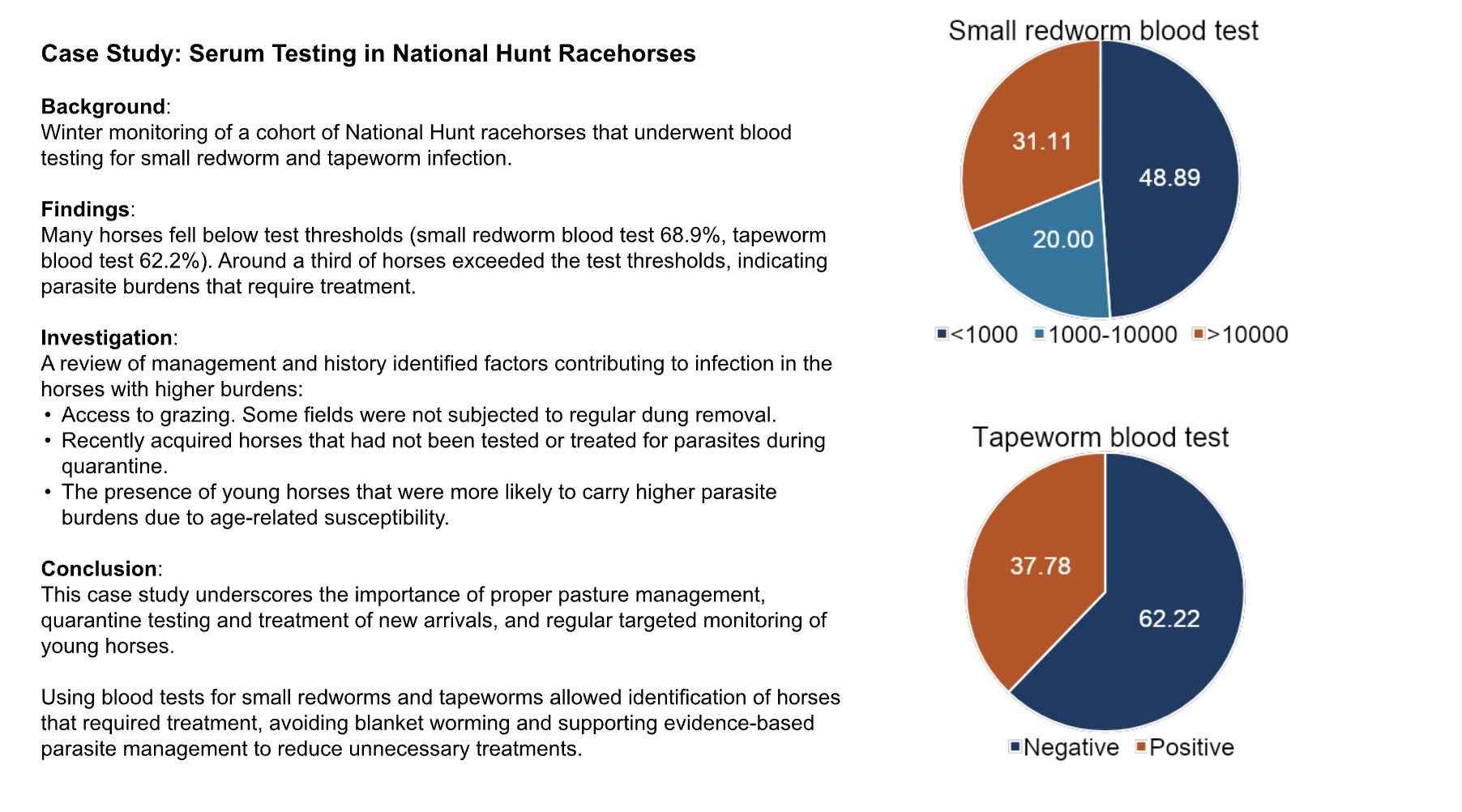
A case study using a testing approach in National Hunt horses is shown in Fig. 3.
Fig. 3
Management approaches
Effective parasite control depends on excellent pasture management to reduce environmental contamination. Key strategies include regular dung removal, maintaining low stocking densities, avoiding overgrazing, and, where suitable, co-grazing with other species. Dung removal is the most effective method as it removes eggs before they develop to infective stages. Removal should be conducted at least twice weekly. Keeping stocking densities to at least one acre/horse helps limit parasite build-up. Resting paddocks for six months or more, or using rotational grazing further reduces infection. Harrowing should be used cautiously. In the UK, conditions are usually not hot or dry enough to kill parasite stages; harrowing merely spreads the worms across paddocks. If harrowing, it is recommended that the paddock remain without horses for a minimum of six months prior to reintroducing them.
Managing new arrivals
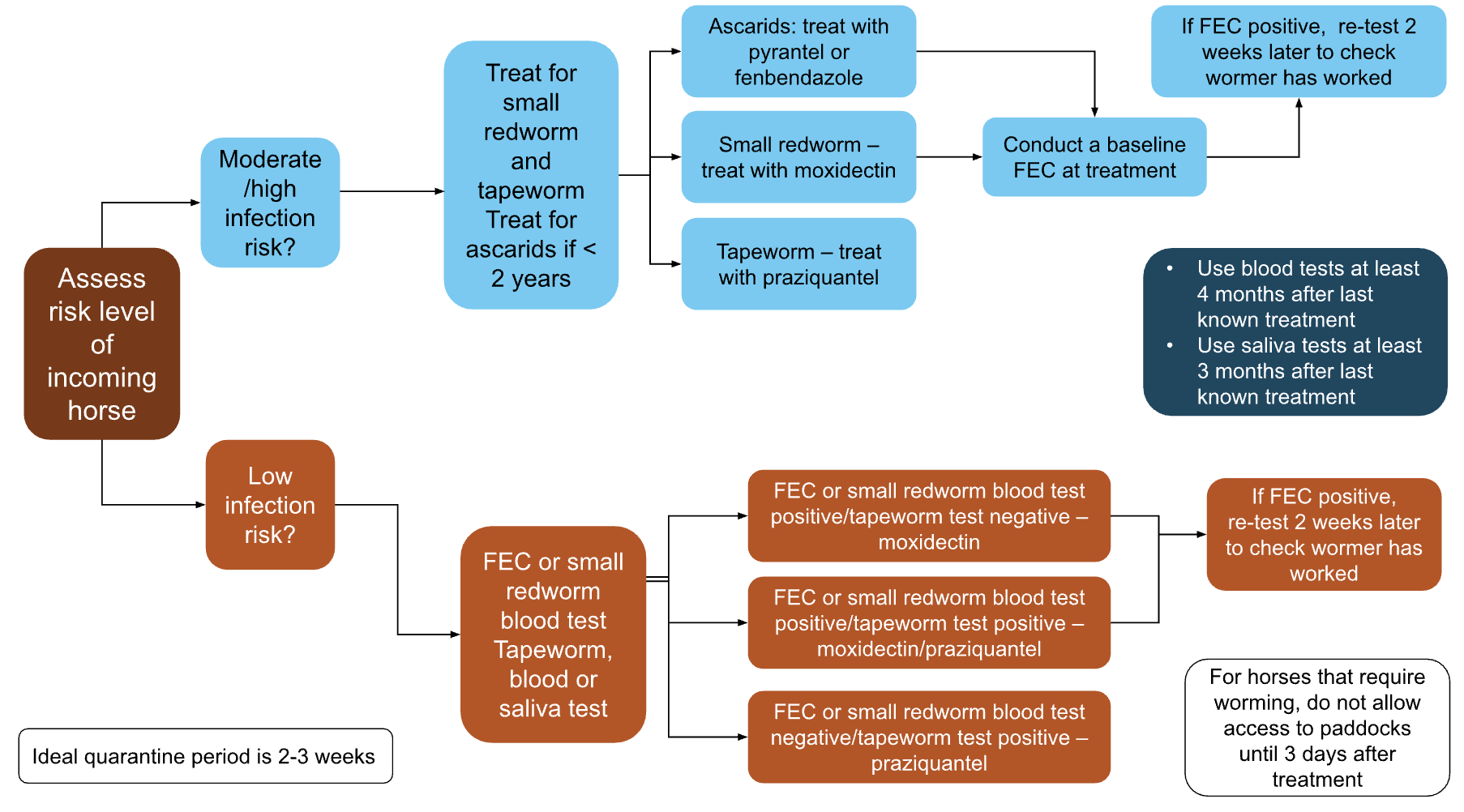
Appropriate quarantine measures for new arrivals are essential to prevent the introduction of new infections or drug-resistant parasites. All new horses should be isolated and tested or treated according to their assessed risk (Fig. 4), and any treatment should be followed up with efficacy testing to assess wormer effectiveness.
Fig. 4
Conclusion
Despite increasing awareness of sustainable parasite control, overuse of wormers remains common, particularly in the Thoroughbred racing sector. A diagnostic-led, risk-based approach, combining targeted testing, careful risk assessment and effective management practices, can reduce reliance on anthelmintics and help preserve their long-term effectiveness.
References
Byrne et al. 2025. Vet Rec Case Rep. 13:e70222.
Lightbody et al. 2024. Int J Parasitol. 54:23-32.
Lightbody et al. 2016. Vet Clin Pathol. 45:335–346.
Matthews et al. 2024. Pathogens. 12:1233.
Matthews et al. 2024. In Practice 46:34-41.
Nielsen. 2022 Int J Parasitol: Drugs Drug Res. 20:76–88.
Nielsen. 2023. Int J Parasitol: Drugs Drug Res. 22:96–101.
Wickenden et al. 2025. Int. J. Parasitol. 14:783-794.